

1 / 5
 Alhilal Healthcare
Alhilal Healthcare










Lymphatic Drainage Massage Machine ECG Holter Monitor Ultrasound System
Model: TLC5000 | Certificate: CE/FDA
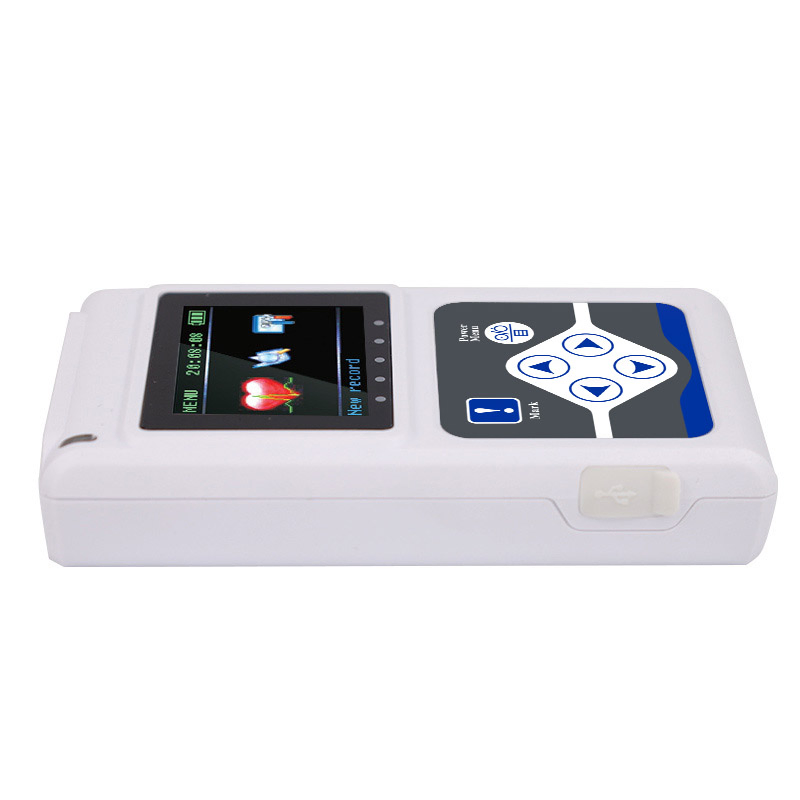
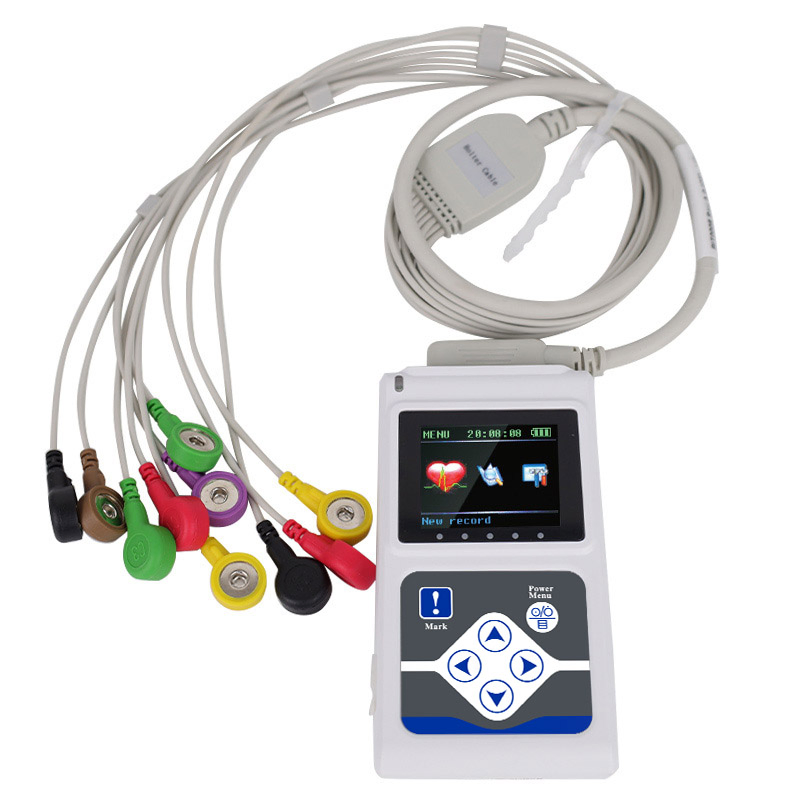
The TLC5000 24-hour Dynamic EKG System is designed to continuously record and analyze ECG waveforms via PC software. Featuring an international standard 12-lead system, it is ideal for medical institutions and community health services.
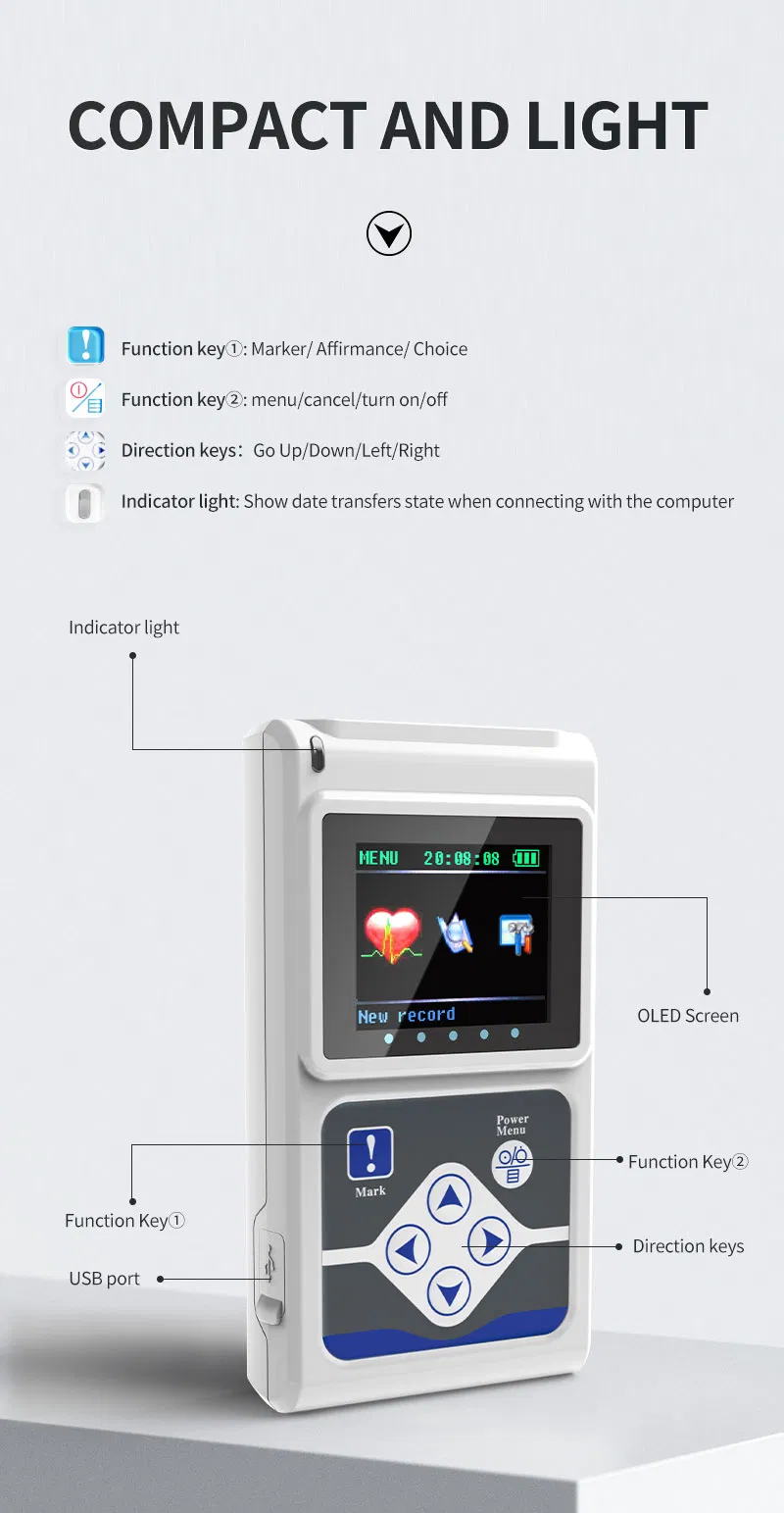


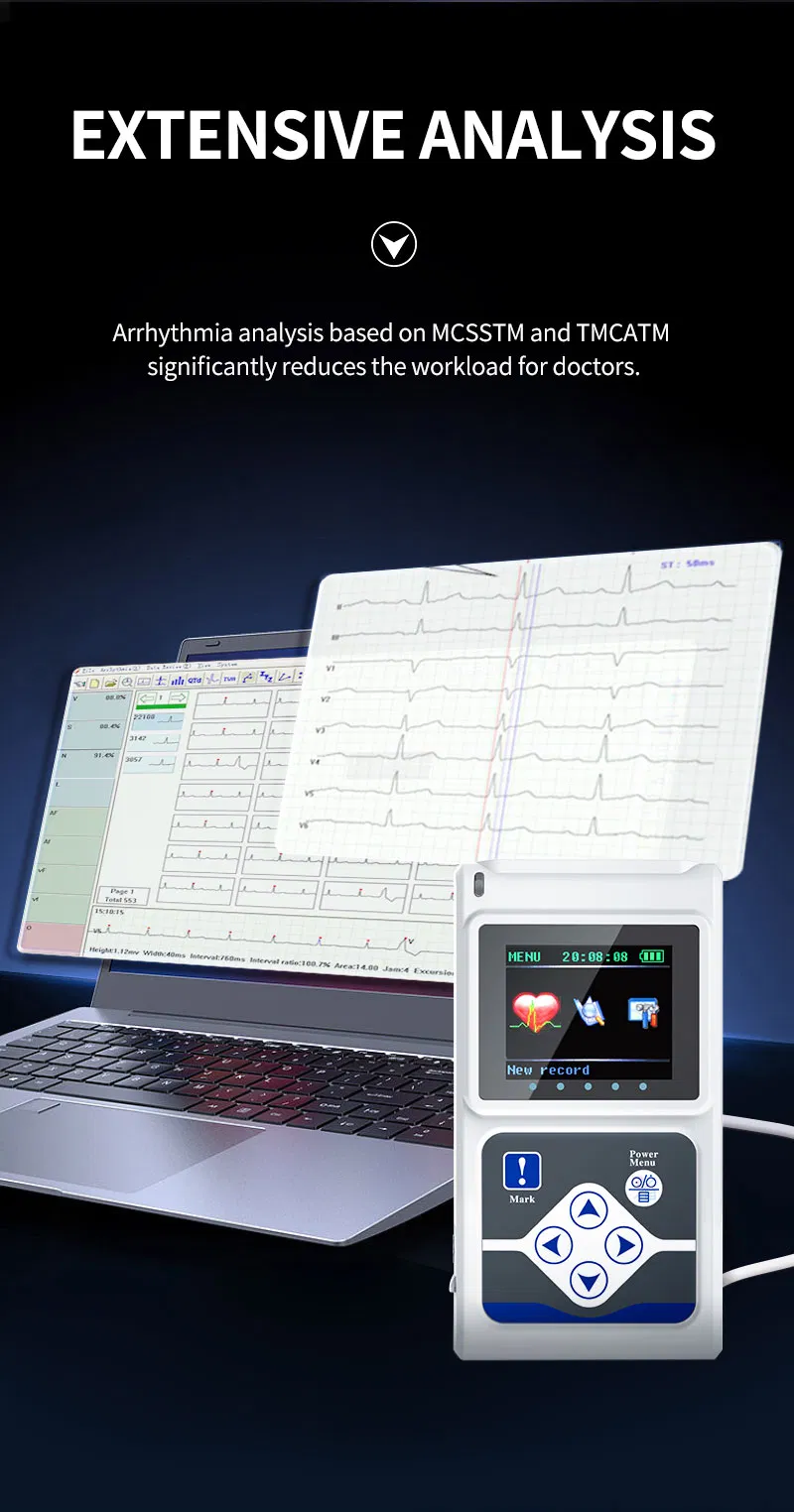
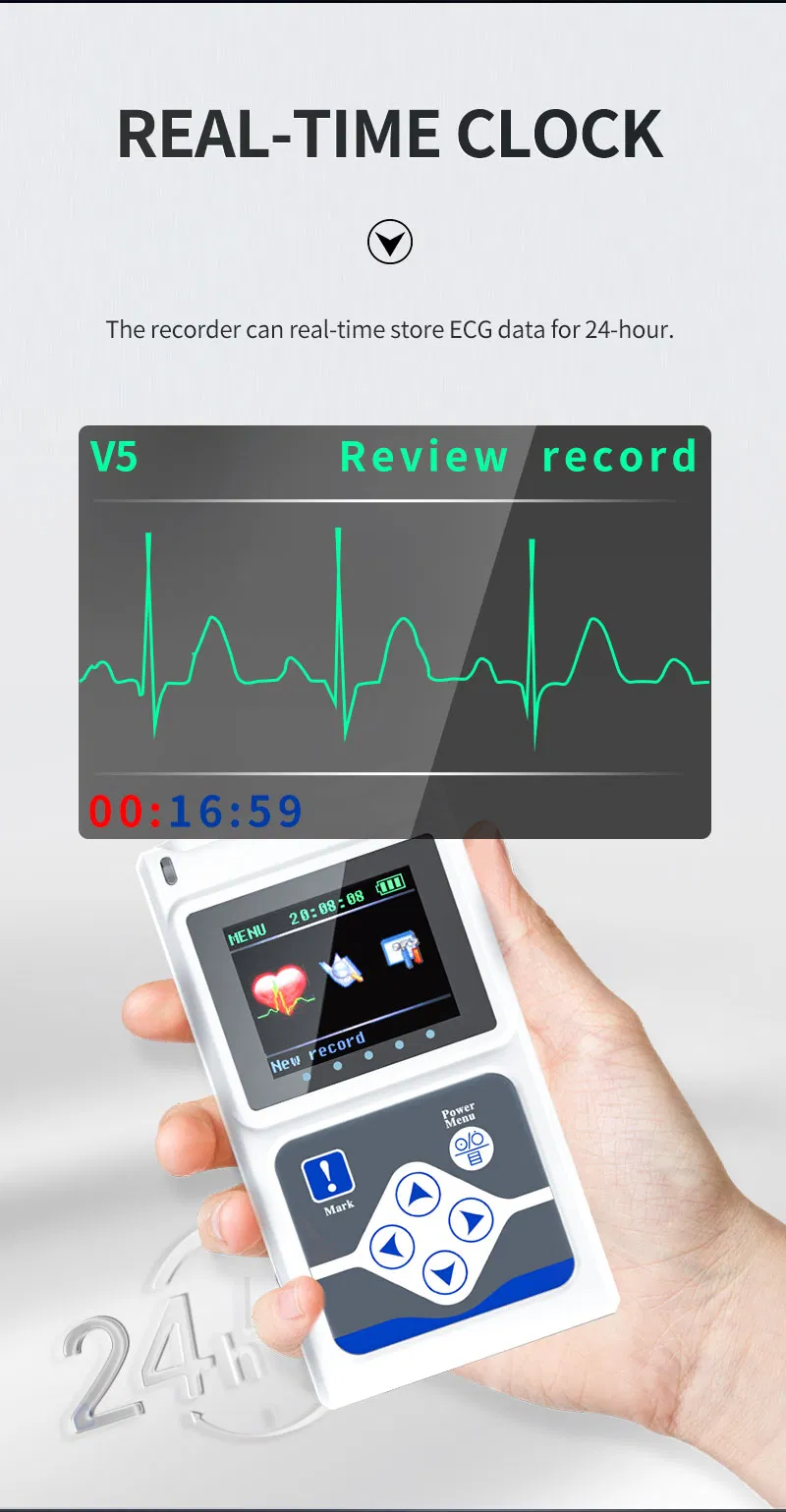
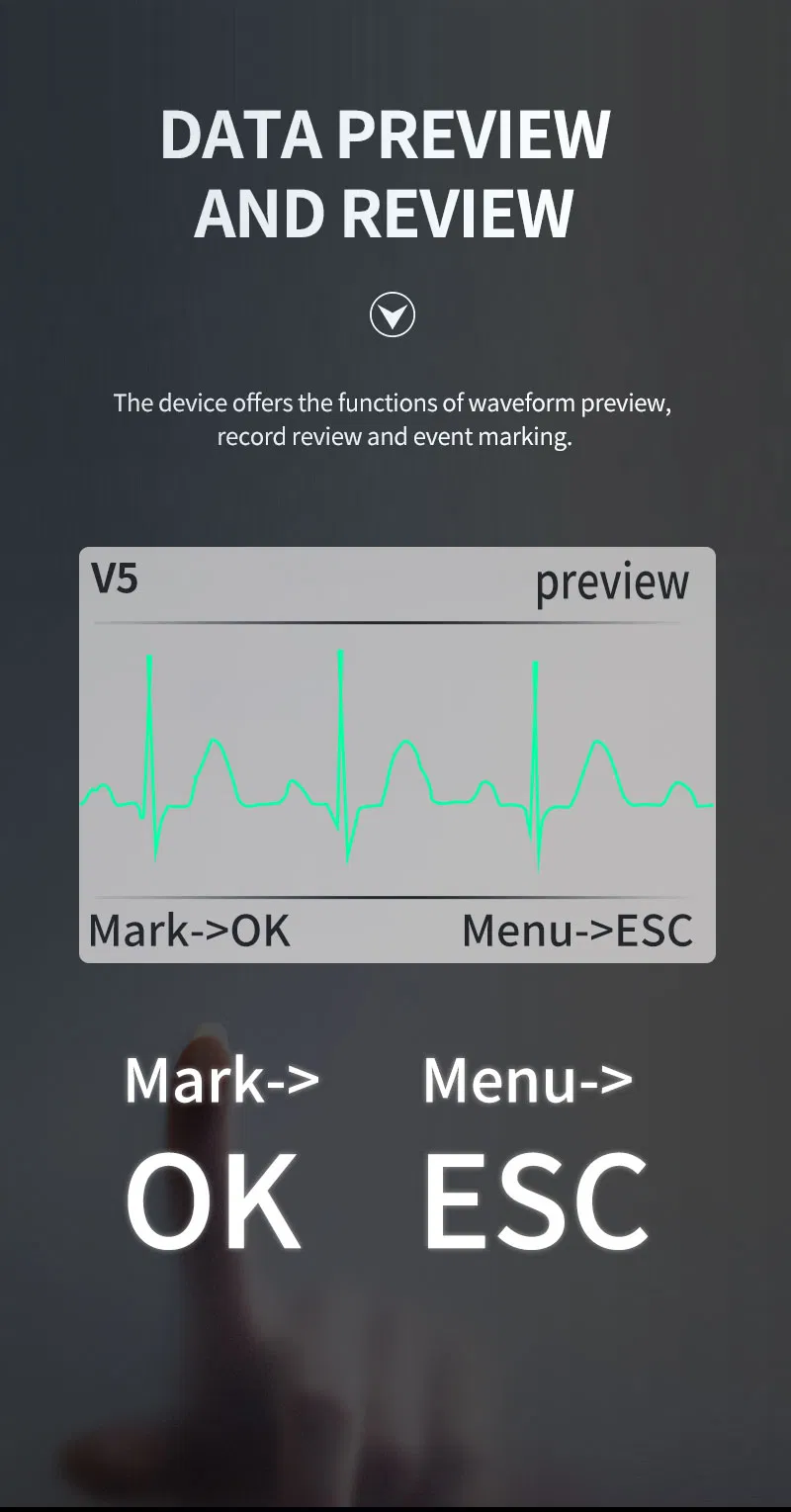
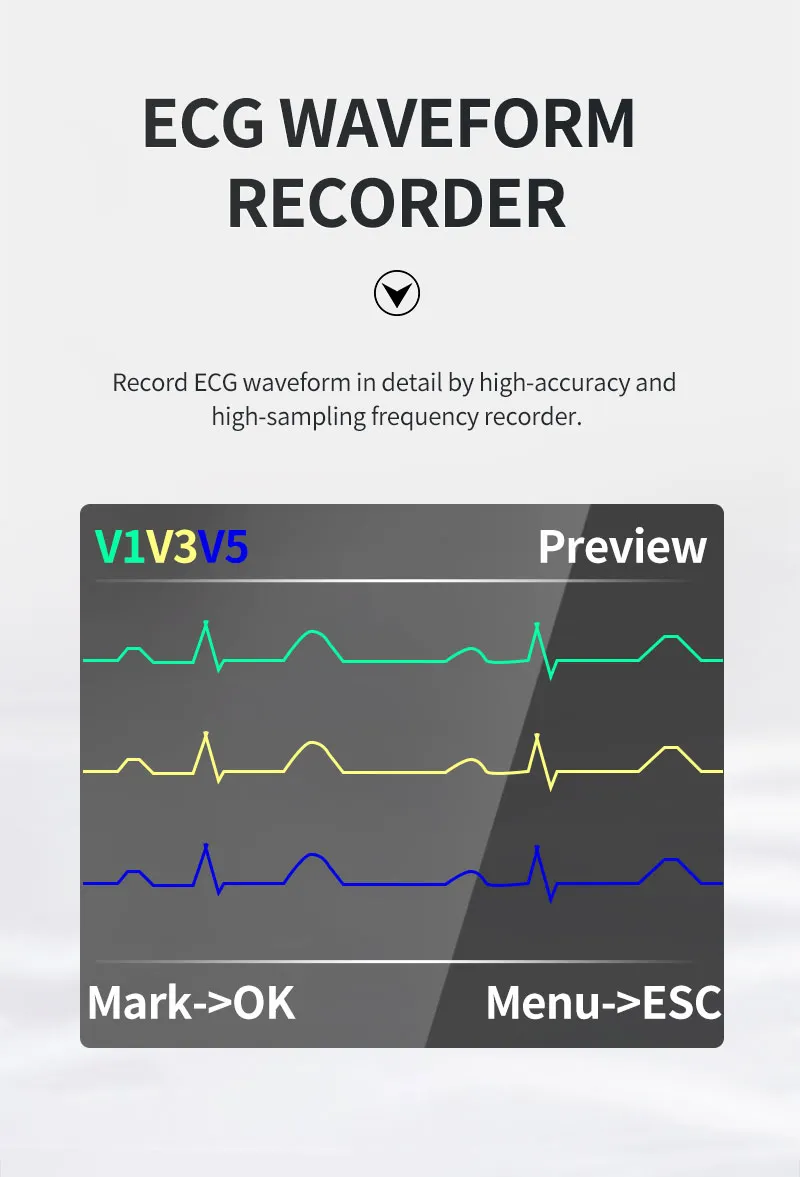
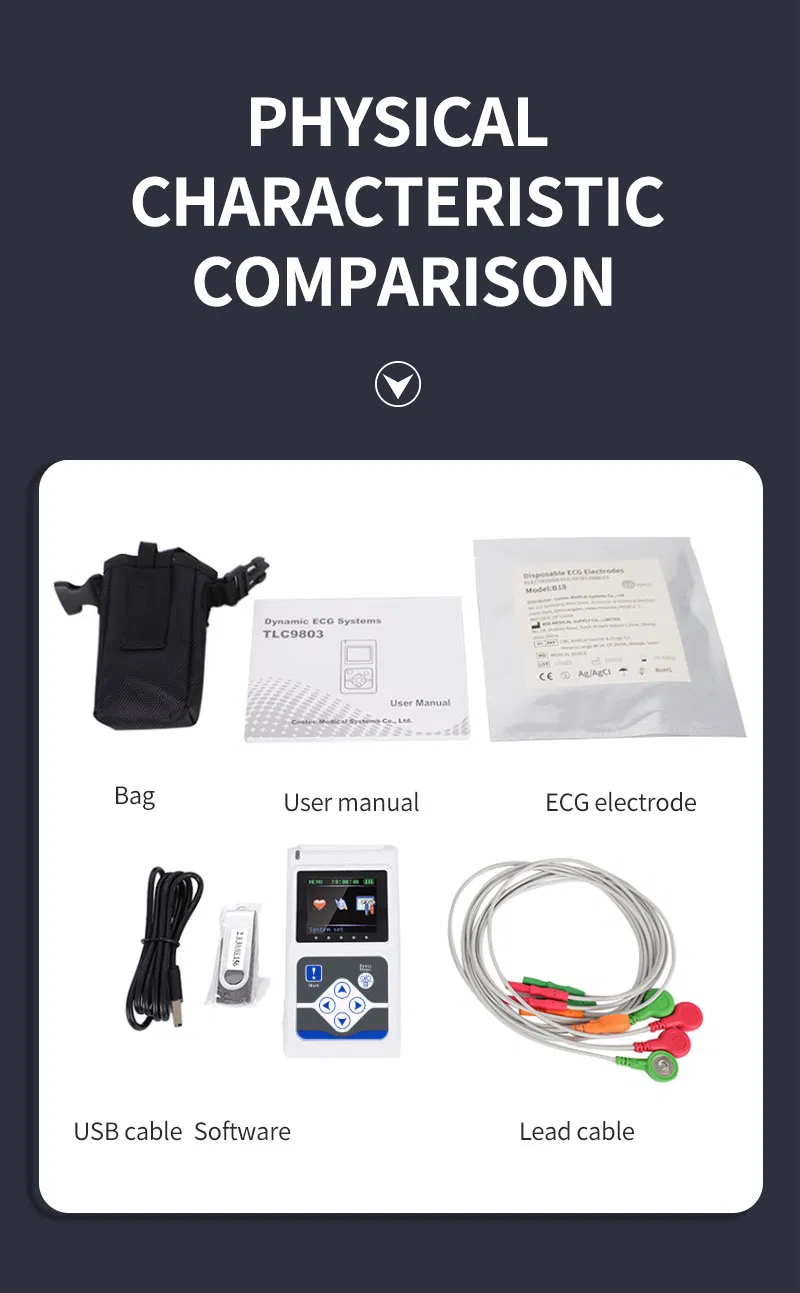
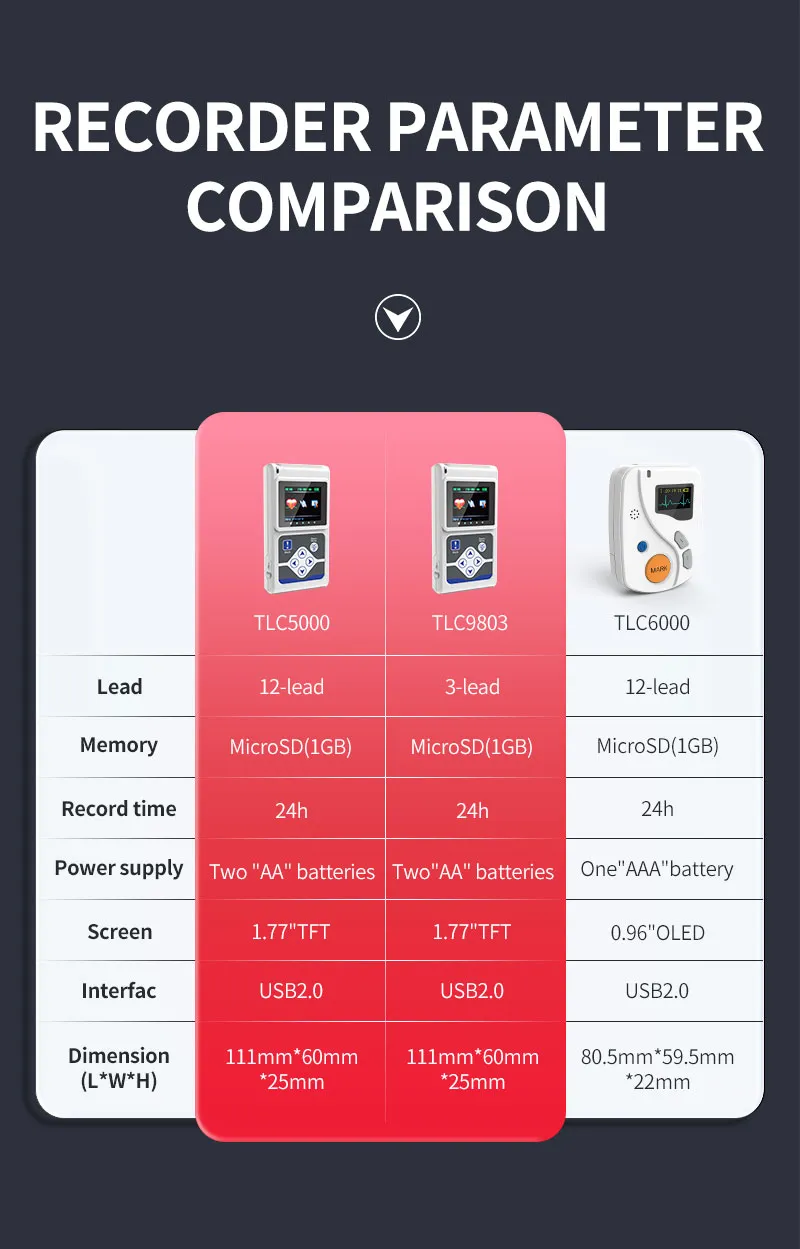
| Lead | Standard 12-lead |
|---|---|
| Record Time | 24-hour |
| Power Supply | Two "AA" batteries |
| Interface | USB 2.0 |
| Calibration Voltage | 1mV ± 5% |
| Noise Level | ≤30μV |
| CMRR | ≥60dB |
| Low-frequency Characteristics | Time constant ≥ 3.2s |
| Scan Speed | 25mm/s ± 5% |
| Polarizing Voltage | ±300mV, sensitivity: ≤±10% |
| Minimum Measure Signal | 50 µV p-p |
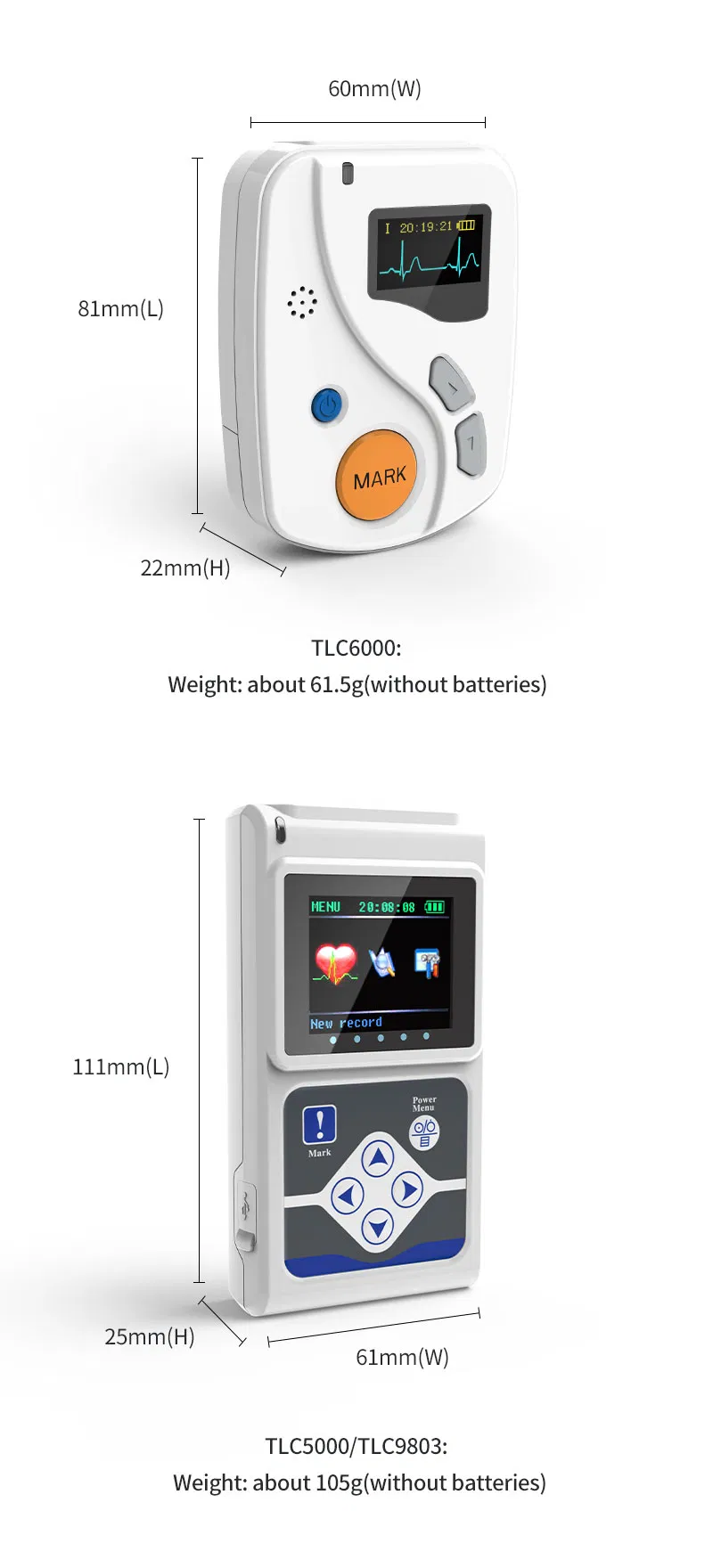
Dimension: 111mm(L) × 60mm(W) × 25mm(H)
Weight: about 105g (without batteries)




