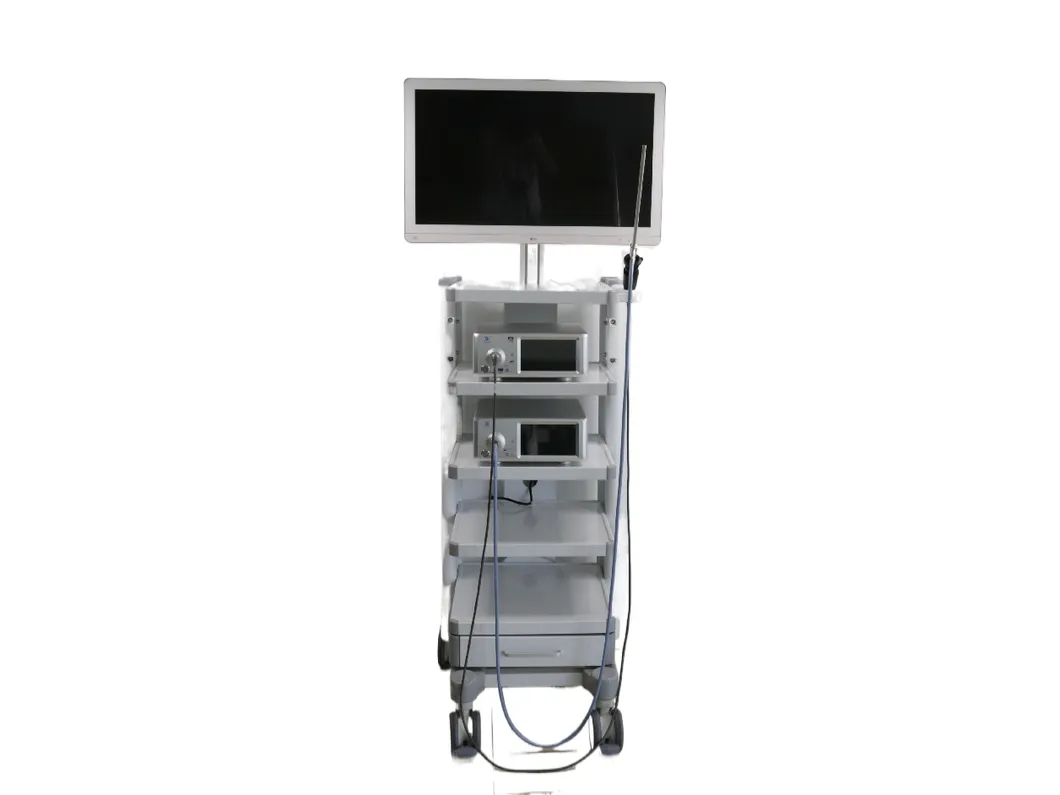
📋 Basic Information
Certification
CE, FDA, EEC, MSDS, ISO13485
Handle Buttons
Awb, Video, Zoom in, Zoom out
Storage Format
Image: JPEG, BMP; Video: AVI
Optional Scene
Laparoscopy, Hysteroscopy, E.N.T, Orthopedics
Resolution
3840X2160p and 1920X1080p
Digital Denoising
High, Medium, Low, and Off Modes
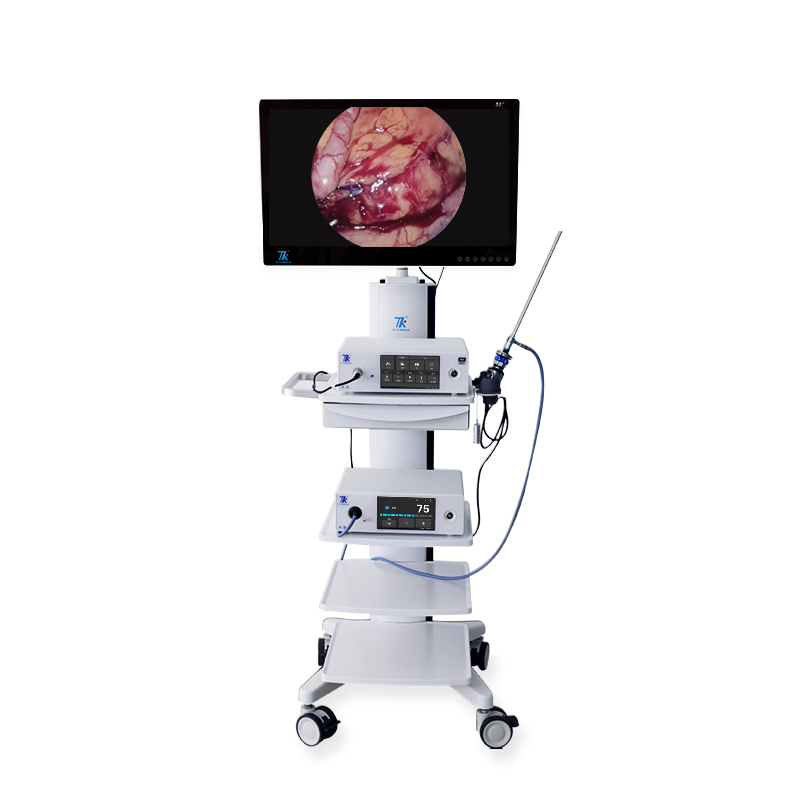
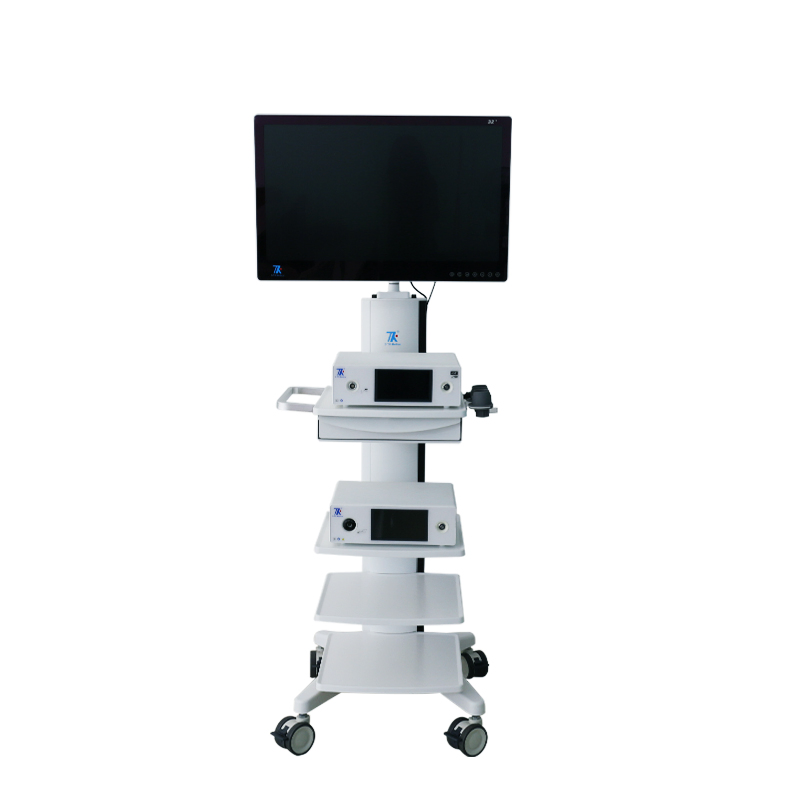
📷 4K Medical Endoscopic Camera System
| Product Name | 4K Medical Endoscopy Camera System |
| Instrument Classification | Class II |
| Screen Size | 32 inches Main Monitor / 7-inch Touch Screen |
| Image Resolution | Ultra HD 3840x2160 (1500TVL) |
| Video Format | Mpeg-4 (H.264 codec) 60fps |
| Storage | SD card / USB Memory |
| Camera Waterproof | IPX8 Rating |
| Connections | 1xHDMI2.0, 1xDVI, 4x12G-SDI |
| Features | Photo/Video, Freeze, Zoom in/out, Auto White Balance |
| Appearance Size | 377X297X137mm |
🏭 Manufacturer Overview
A high-tech enterprise specializing in medical devices since 2004, dedicated to the research, development, and production of minimally invasive surgical instruments and novel oral care products. Operating from a modern factory spanning over 10,000+ square meters, the team includes 140+ medical industry professionals. With over 700+ domestic and international patents, the company provides advanced medical solutions to over 40+ countries worldwide.
🌐 International Exhibitions
❓ Frequently Asked Questions
Are you a manufacturer or a trading company?
We are a dedicated manufacturer in China with 20 years of experience in developing and producing surgical devices.
What are the core technical advantages of your systems?
Our strengths lie in our robust R&D capabilities, with over 600 applied patents and world-class quality management holding FDA 510K, CE, and ISO13485:2016 certifications.
What is the image quality of the endoscopic camera?
The system provides Ultra HD imaging with a resolution of 3840x2160p (1500 TV Lines), ensuring crystal clear visuals for surgical procedures.
Has the equipment been exported to international markets?
Yes, our products are exported to more than 40 countries, with a primary focus on the North American and European medical markets.
Do you accept factory audits from international partners?
Yes, we are confident in our manufacturing standards and regularly pass on-site audits, including US FDA audits in 2015 and 2021.
What clinical applications is this system suitable for?
It is designed for various minimally invasive surgeries, including Laparoscopy, Hysteroscopy, E.N.T., Orthopedics, and Fiber-optic endoscopies.



 Alhilal Healthcare
Alhilal Healthcare